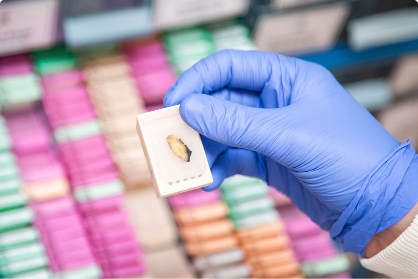
Join the Nottingham BioResource
The Nottingham BioResource is a “bank” of samples and data from people with common and rare diseases, as well as healthy members of the public.
The samples are donated by people who want them to be available for use in clinical research. The samples we collect from volunteers are often the samples you would give as part of your routine treatment and care – a sample of blood or urine, for example.

Donating samples
Any member of the public is eligible to donate samples to the Nottingham BioResource service. Samples may include:
- Blood
- Bodily fluids – for example a urine sample
- Tissue
- Data – information about their health usually from their health record
Any member of the public is eligible to donate samples to the Nottingham BioResource service. Samples may include:
To donate you can:
- Speak to the team caring for you at NUH
- A member of the team from the BioResource service will discuss your donation with you
- You will be given information about the BioResource and how your sample(s)/data can be used by researchers
- You will be asked to sign a document to show that you agree (consent)
- You will also be told about the process if you change your mind at any time
Donating your samples to the Nottingham BioResource service is voluntary. Your decision will not affect the treatment and care you receive from NUH.
The Human Tissue Act
The Nottingham BioResource service operates under the Human Tissue Act 2004. This is a legal framework which regulates the “removal, storage, use and disposal of human bodies, organs and tissues”.
The Act came into effect on the 1st September 2006 and applies to England, Wales and Northern Ireland.
The Nottingham BioResource is a Human Tissue Authority (HTA) licensed facility.
Human biological samples for research must be traceable from collection through to disposal.
Ethics:
The work the Nottingham BioResource does is reviewed and approved by an independent Research Ethics Committee (RECs).
The role of Research Ethics Committees is to decide if research is ethical. They are part of a framework that regulates research, under the NHS Health Research Authority.
The Nottingham BioResource has the below ethics approval in place:
- Research Tissue Bank (REC REF: XX/XX/00XX)
- Rare Diseases (REC REF: XX/XX/00XX)
Get involved
Requests to use samples and data from the Nottingham BioResource are considered by an expert Committee. Our Committee involves members of the public and patients who work with our panel of experts to consider these requests.
If you would be interested in volunteering to be a member of the Access Committee, please contact us
You can find out about other volunteering opportunities in clinical research in Nottingham on the Research Hub [hyperlink to Research Hub].
Nottingham’s biomedical database and research resource

Using our services

Enquire about samples